Virgile VIASNOFF
Associate Professor, Mechanobiology Institute, National University of Singapore
Virgile.Viasnoff@espci.fr
09-03-05
Level 9 T-Lab
National University of Singapore
5A Engineering Drive 1
Singapore 117411
Laboratory website
Biomechanics of Cell-Cell Contacts
Research Program
Technology Innovation for Mechanobiology
Affiliations
Charge de Recherche 1st Class CNRS/ France
Associate Professor, Department of Biological Sciences, National University of Singapore
The Crown JeWell of organoid imaging
An interdisciplinary team from MBI combined imaging, microfabrication, and biology to develop JeWells - an innovative platform for growing and imaging organoids in 3D. Learn more
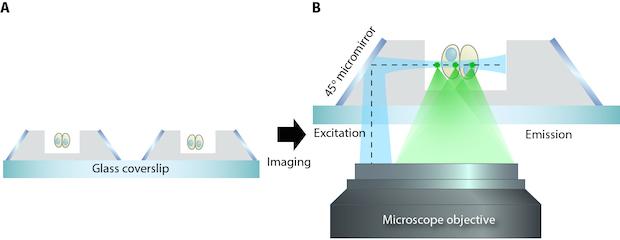
So-SPIM-FCS
A novel tool for imaging nuclear protein dynamics
B(a)iled Out
Actin remodeling drives bile regurgitation during obstructive cholestasis
Shaping Lumens by Force
How extracellular matrix scaffolding guides lumen elongation
soSPIM Developed at MBI
Microfabrication leads to a new microscopy method
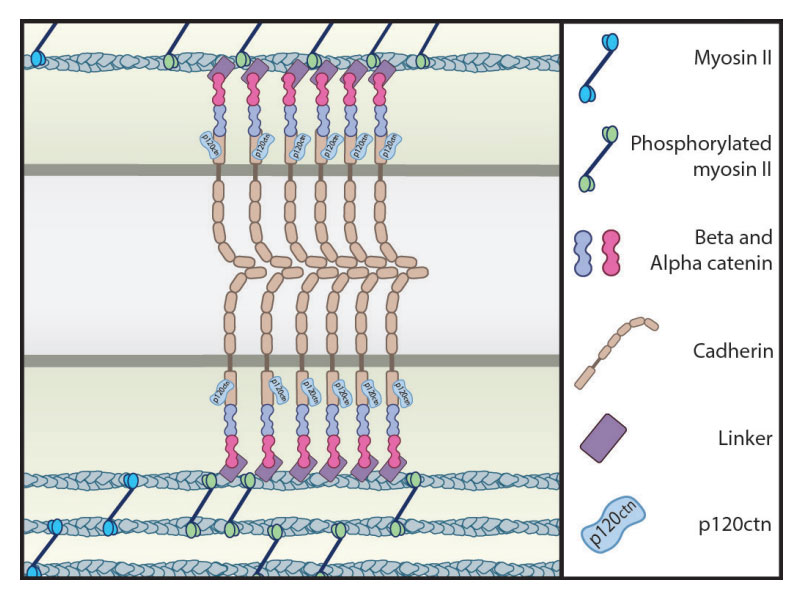
Capturing E-cadherin
Dynamics of cortical actin in the recruitment o cadherin at adhesion sites
Virgile Viasnoff
Principal Investigator
Research Areas
Cell-cell adhesion, microfabrication of controlled environment for cell culture, biophysic of single molecules
Research Interests
Cell-cell adhesion is a fundamental biological process in tissue formation, embryogenesis and cancer development. Our research aims at understanding the physical and biomolecular mechanisms that underlies the formation of cell-cell junction in various conditions. One approach that we have taken is to use model cell systems to understand the dynamics and spatial organization of cadherin during the formation of the junction. We also develop physical approaches to test and measure the influence of mechanical tensions on the structure and dynamics of junction formation. Another approach is to study specific junction such as bile canaliculi formed between primary hepatocytes . Using microfabricated patterns with 3D protein coating we control the dynamics and position of bile canaliculi formation. Performing specifically developed techniques we image in real time the formation of the secretion tube and study how its formation is affected by physical parameters.
Building on our experience with 3D protein coating in nano and microtextured environment, we are developing a platform for controlled cell culture, high resolution imaging and high through-put screening capabilities.
Biography
Virgile Viasnoff was trained as a soft matter physicist at Ecole Normale Superieure of Paris. After one Year at Johns Hopkins (USA) as a master thesis, he joined ESPCI (Paris) where he did his PhD on the microrheology of aging colloidal system. He then spent a 2 year post doc in Harvard where he worked on nanopore technologies to study RNA folding kinetics. In 2006 he was recruited at CNRS at ESPCI in Paris where he now works on single molecule biophysics. In parallel to his activity in Paris, he joined MBI in 2010 as an Associate Prof where he started working on cell-cell junction mechanics and microfabrication of patterned substrate for cell culture. He is now heading a collaborative Research program between MBI and CNRS on cell adhesion.
Education
PhD ESPCI (Paris)
Recent Publications
- Harley P, Paredes-Redondo A, Grenci G, Viasnoff V, Lin Y, and Lieberam I. 3D Compartmentalised Human Pluripotent Stem Cell-derived Neuromuscular Co-cultures. Bio Protoc 2023; 13(5):e4624. [PMID: 36908638]
- Grenci G, Dilasser F, Mohamad Raffi SB, Marchand M, Suryana M, Sahni G, Viasnoff V, and Beghin A. A High-Throughput Platform for Culture and 3D Imaging of Organoids. J Vis Exp 2022;(188). [PMID: 36314813]
- Kai F, Ou G, Tourdot RW, Stashko C, Gaietta G, Swift MF, Volkmann N, Long AF, Han Y, Huang HH, Northey JJ, Leidal AM, Viasnoff V, Bryant DM, Guo W, Wiita AP, Guo M, Dumont S, Hanein D, Radhakrishnan R, and Weaver VM. ECM dimensionality tunes actin tension to modulate endoplasmic reticulum function and spheroid phenotypes of mammary epithelial cells. EMBO J 2022;:e109205. [PMID: 35880301]
- Beghin A, Grenci G, Sahni G, Guo S, Rajendiran H, Delaire T, Mohamad Raffi SB, Blanc D, de Mets R, Ong HT, Galindo X, Monet A, Acharya V, Racine V, Levet F, Galland R, Sibarita J, and Viasnoff V. Automated high-speed 3D imaging of organoid cultures with multi-scale phenotypic quantification. Nat Methods 2022;. [PMID: 35697835]
- Chew EGY, Lim TC, Leong MF, Liu X, Sia YY, Leong ST, Yan-Jiang BC, Stoecklin C, Borhan R, Heilmann-Heimbach S, Nöthen MM, Viasnoff V, Shyh-Chang N, Wan ACA, Philpott MP, and Hillmer AM. Observations that suggest a contribution of altered dermal papilla mitochondrial function to androgenetic alopecia. Exp Dermatol 2022;. [PMID: 35119146]
- Fu C, Arora A, Engl W, Sheetz M, and Viasnoff V. Cooperative regulation of adherens junction expansion through Epidermal Growth Factor Receptor (EGFR) activation. J Cell Sci 2022;. [PMID: 35099018]
- Aladin DMK, Chu YS, Shen S, Robinson RC, Dufour S, Viasnoff V, Borghi N, and Thiery JP. Extracellular domains of E-cadherin determine key mechanical phenotypes of an epithelium through cell- and non-cell-autonomous outside-in signaling. PLoS One 2021; 16(12):e0260593. [PMID: 34937057]
- Nishimura Y, Shi S, Li Q, Bershadsky AD, and Viasnoff V. Crosstalk between myosin II and formin functions in the regulation of force generation and actomyosin dynamics in stress fibers. Cells Dev 2021;:203736. [PMID: 34455135]
- Nishimura Y, Shi S, Zhang F, Liu R, Takagi Y, Bershadsky AD, Viasnoff V, and Sellers JR. The Formin Inhibitor, SMIFH2, Inhibits Members of the Myosin Superfamily. J Cell Sci 2021;. [PMID: 33589498]
- Lenne P, Rupprecht J, and Viasnoff V. Cell Junction Mechanics beyond the Bounds of Adhesion and Tension. Dev Cell 2021;. [PMID: 33453154]
Lab Members
Modhura Ganguly
PhD Student, Class of January 2022, Viasnoff Group
Marion Florence Marchand
Research Fellow, Viasnoff Group
Deepika Singh
PhD Student, Class of August 2019, Viasnoff Group
Harini Rajendiran
Research Assistant, Viasnoff Group