How Cells Repair Wounds
Steven J Wolf | August 2014
A collaborative study between the Mechanobiology Institute, Singapore (MBI), the Institut de Bioenginyeria de Catalunya, Paris Diderot University, the IRB, UPC and UB in Barcelona, and the University of Waterloo in Canada has found a new way to decipher the mechanisms of wound healing. This research has provided a new understanding of how cells move and work together to close gaps. This work, published today in Nature Physics, signifies a big step forward in unravelling the mystery of how wounds are repaired and could eventually assist in the development of drugs that enhance or quicken healing.
Agustí Brugués, Ester Anon, Vito Conte, Jim H. Veldhuis, Mukund Gupta, Julien Colombelli, José J. Muñoz, G. Wayne Brodland, Benoit Ladoux, Xavier Trepat. Forces driving epithelial wound healing, Nature Physics, epub ahead of print (2014).
Collaborative study identifies new mechanism for wound healing
The stinging or burning sensation that accompanies a scratch or graze is an all too familiar feeling. What we fail to appreciate is that almost immediately after we feel that sting, an intricate mechanism is initiated that sees cells begin to heal the wound. Importantly, wounds may also occur deep inside the body in any tissue or organ, and these can lead to the development of chronic diseases such as diabetes and asthma. In some cases they may encourage the onset of cancer by providing a physical and chemical environment that promotes cancer cell invasion.
Now, a collaborative study between the Mechanobiology Institute, Singapore (MBI), the Institut de Bioenginyeria de Catalunya, Paris Diderot University, the IRB, UPC and UB in Barcelona, and the University of Waterloo in Canada has found a new way to decipher the mechanisms of wound healing. This research has provided a new understanding of how cells move and work together to close gaps. This work, published today in Nature Physics, signifies a big step forward in unravelling the mystery of how wounds are repaired and could eventually assist in the development of drugs that enhance or quicken healing.
A new mechanism of wound healing
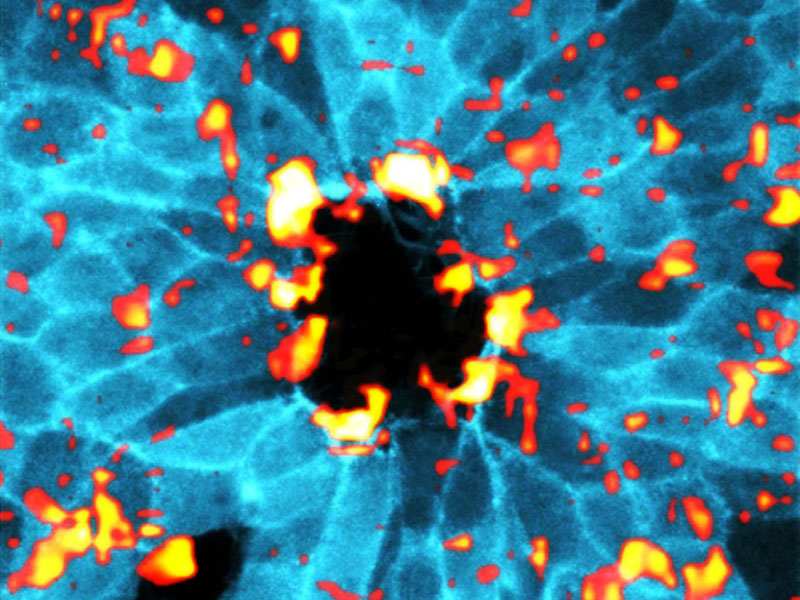
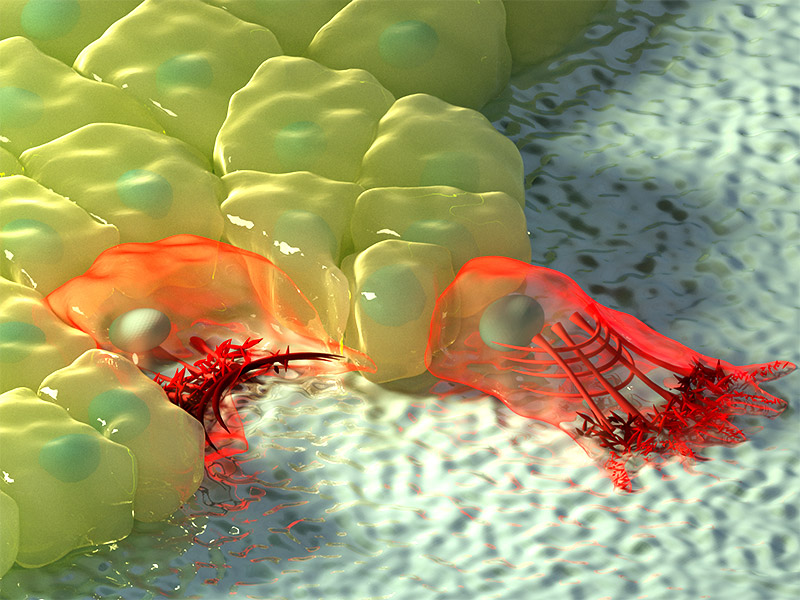
It is well established that two different mechanisms contribute to wound healing. One is the ‘purse-string’ mechanism, where a ring of contractile proteins forms at the edge of a wound and tighten like the strings of a purse. The other mechanism is ‘cell crawling’, where cells reach out using arm-like projections known as filopodia and lamellipodia, to drag themselves forward in order to close the gap. In some wounds, both mechanisms are thought to occur simultaneously, while in others only one of the two is initiated.
Ladoux, Trepat and colleagues have pioneered a technique to measure the nano-scale forces behind wound healing and, in doing so, have discovered that the two currently accepted mechanisms are insufficient to fully explain the phenomenon of wound healing. Instead, they have revealed a new mechanism, whereby cells assemble supracellular-contractile arcs that compress the tissue underneath the wound.
New research signifies a big step forward in unravelling the mystery of how wounds are repaired and could eventually assist in the development of drugs that enhance or quicken healing.
By combining experiments and computational modeling, the authors have shown that contractions arising from these arcs speed up wound healing in a more robust manner. The direct measurement of cell-generated forces has shed new light on our understanding of wound healing processes. It reveals that cellular contractile forces and their impact on the underlying substrate, could help to increase wound healing efficiency.
This study shows that the interplay between cell-generated forces and the deformation of the underlying substrate is crucial to explain wound healing mechanisms and more generally, could play a major role in collective cell movements. Interestingly, in vivo, the underlying substrate could be either extracellular matrix or other tissues whose deformation may guide large-scale cellular movements and help for an efficient tissue remodelling.
Being able to optimize tissue repair is needed for the treatment of acute and chronic diseases. The discovery of the basic mechanism reported in this study clearly underlines the importance of cellular mechanics in understanding major biological functions.